Xenemetrix Ltd- The power to change Energy into Information

NEW !!! FUEL INTEGRITY PROGRAM
Governments and oil companies around the world lose significant amounts of revenue every year as a result of the illegal diversion of low-grade or tax-free transit fuels into premium grade domestic fuels. Fuel integrity programs from multiple companies and integrators around the world offer a reliable and cost-effective way of combating this kind of illegal activity, bringing benefits to governments, the oil industry and consumers alike.
Our systems support both existing programs as well as new to come programs.
 Governments and oil companies around the world lose significant amounts of revenue every year as a result of the illegal diversion of low-grade or tax-free transit fuels into premium grade domestic fuels. Fuel integrity programs from multiple companies and integrators around the world offer a reliable and cost-effective way of combating this kind of illegal activity, bringing benefits to governments, the oil industry and consumers alike.
Governments and oil companies around the world lose significant amounts of revenue every year as a result of the illegal diversion of low-grade or tax-free transit fuels into premium grade domestic fuels. Fuel integrity programs from multiple companies and integrators around the world offer a reliable and cost-effective way of combating this kind of illegal activity, bringing benefits to governments, the oil industry and consumers alike.
Our systems support both existing programs as well as new to come programs.
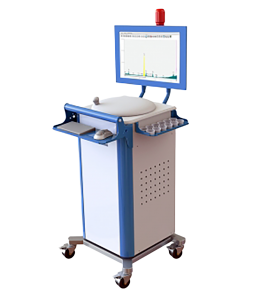
With more than 30 years experience, Xenemetrix Ltd. specializes in the design, development, production and marketing of Energy-Dispersive X-Ray Fluorescence (ED-XRF) systems.
Make an enquiry






call now
Xenemetrix's highly innovative technologies
Xenemetrix continues to develop highly innovative XRF technologies and solutions suitable for today’s ever-growing analytical challenges, performing non-destructive elemental analysis from C(6) to Fm(100), while providing detection limits from low sub-ppm (parts per million).









Applications

Petroleum & Petrochemical
Oil and fuel analysis – compliance with ASTM standard methods and other regulations